Review written by Jarome Ali (EEB, G4)
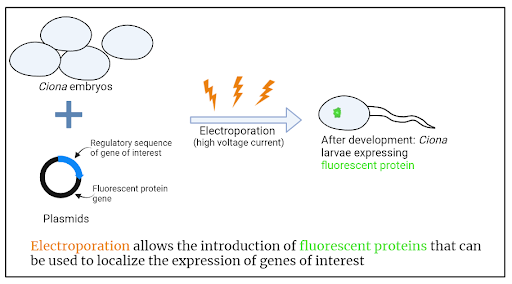
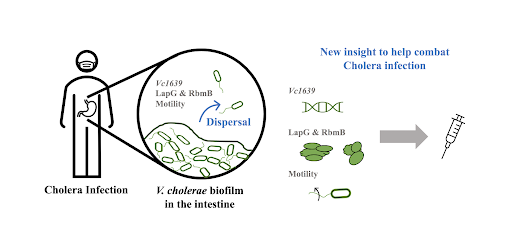
Clinging to the side of a marina you might find the usual suspects like mussels and barnacles, but lurking among these life forms is a less familiar one, a squishy invertebrate that looks like nothing more than a translucent tube. This creature is the sea squirt, Ciona instestinalis. Despite its unassuming appearance, Ciona could hold the key to understanding how the brain--the most complex structure in the universe--came to be. By investigating the evolutionary origin of the brain, we can uncover the roots of the remarkable variety of intelligence in the animal world and gain a deeper appreciation for the beautifully complex human brain. Princeton researchers are now studying the simple ‘brain’ of the sea squirt to begin to unravel this evolutionary story.
Continue reading