Review written by Laura W. Hirschfield, UG ‘24 (Neuroscience)
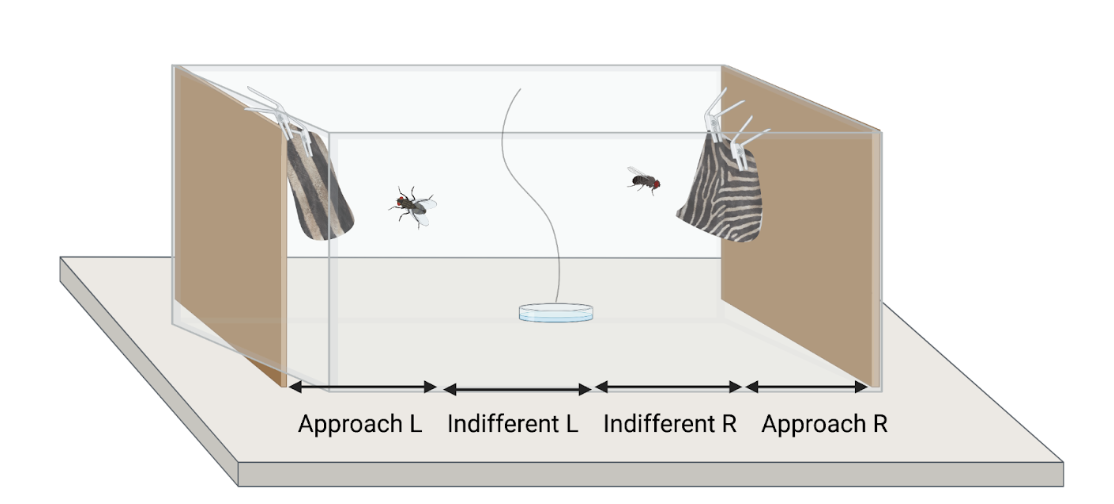
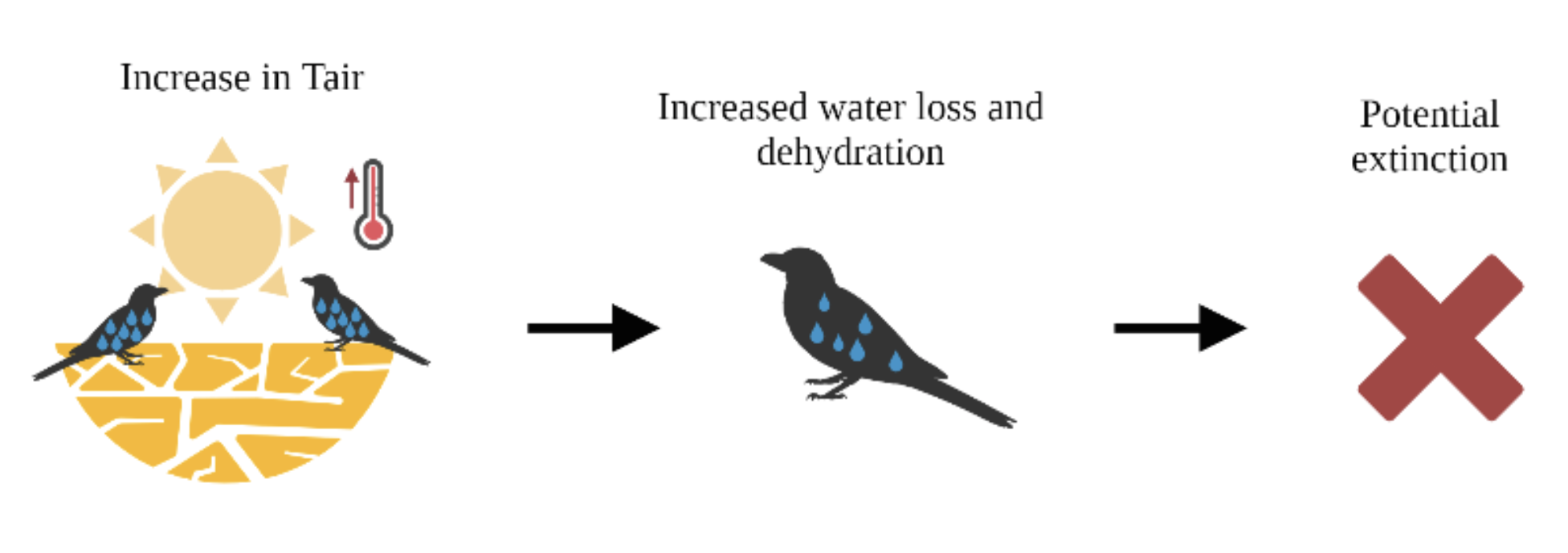
Scientists have continued to debate for centuries the reasons behind why the zebra got its stripes. The zebra’s pattern is rare in the animal kingdom, intensely expressed in both sexes, conspicuous, and yet, its utility is not apparent. Unlike the many other hoofed mammals, called ungulates, that also occupy the east African savannah, the stripes do not provide camouflage for any of the habitats zebras occupy.
Continue reading “Why the Zebra Got its Stripes: exploring the anti-parasite hypothesis”