Review written by Aishwarya Mandyam (COS, G1)
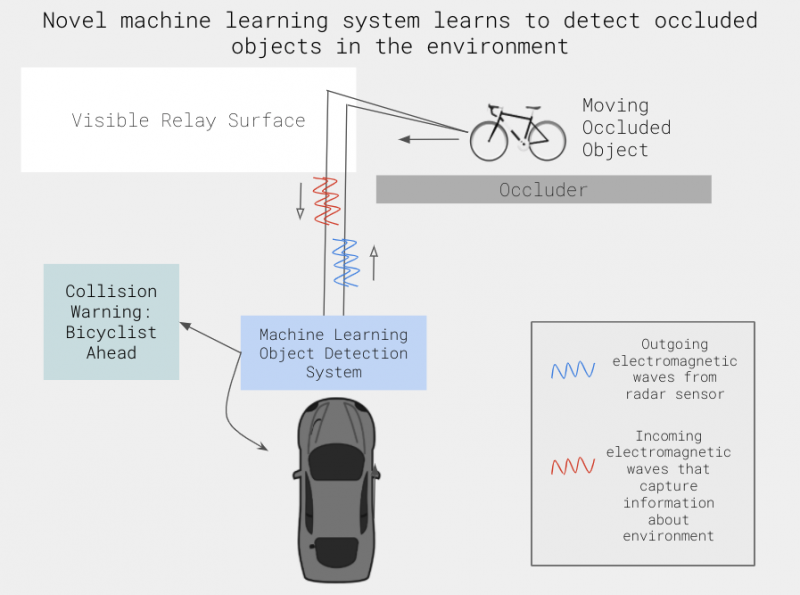
The prevalence of self-driving or partially self-driving cars is increasingly within our sights. However, before these vehicles can make their mainstream debut, it’s important that consumers are ensured of their safety. In particular, these cars should be able to “see” and “act” like human drivers. As such, a self-driving car’s collision and error detection system must effectively reason about what it cannot see. A big concern with self-driving cars is the question of safety; we expect that a human driver has sufficient intuition to avoid pedestrians and stationary objects. Naturally, we would expect self-driving cars to exhibit the same level of caution for avoiding collisions. In a new paper out of Professor Felix Heide’s group in the Princeton COS department in collaboration with Mercedes Benz, Scheiner et al. introduce a method that uses Doppler radar to enable cars to see around corners. Scheiner et al.’s method to track hidden objects allows for a more effective error detection method that can make these vehicles better suited to operating in the real world where safety is a primary concern.
Continue reading