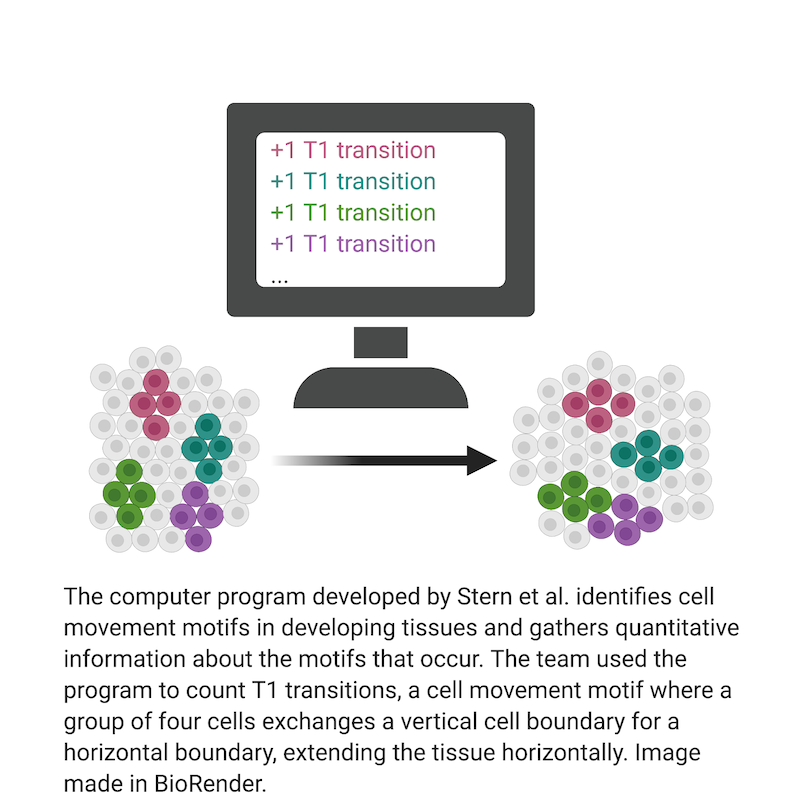
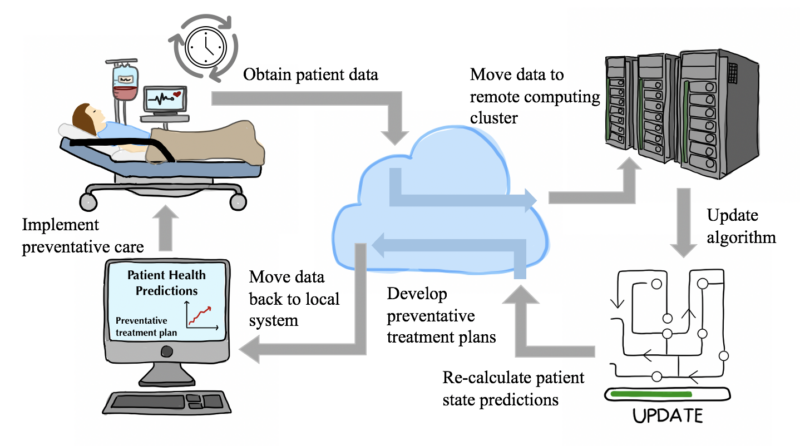
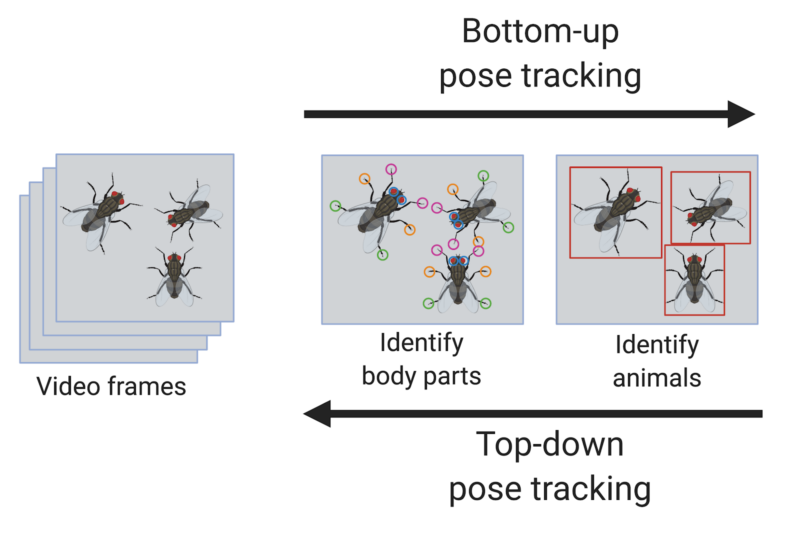
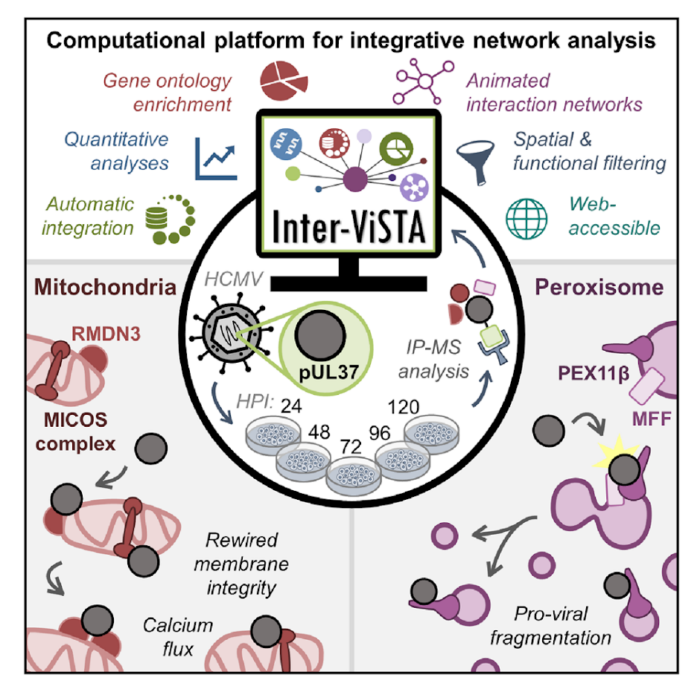
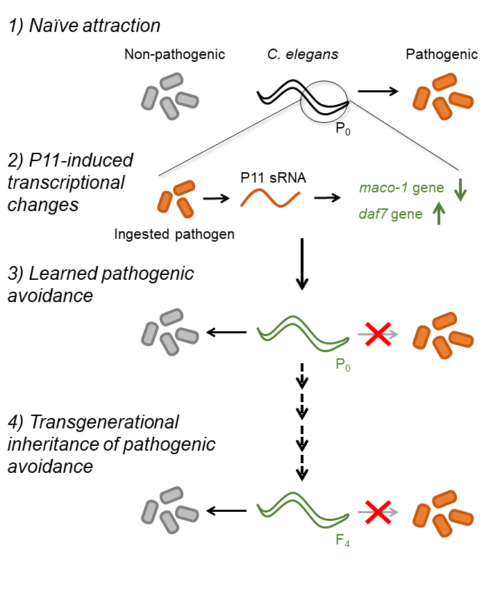
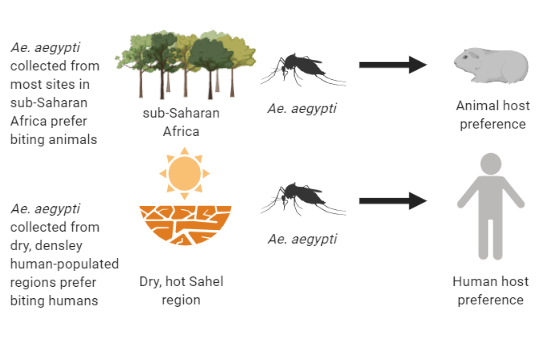
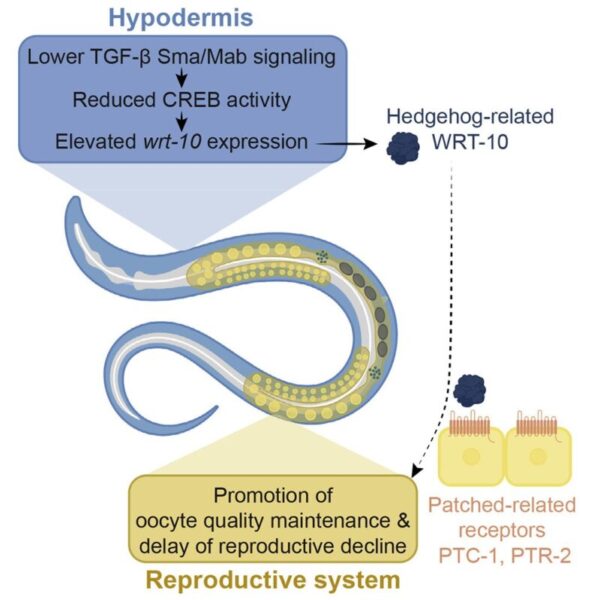
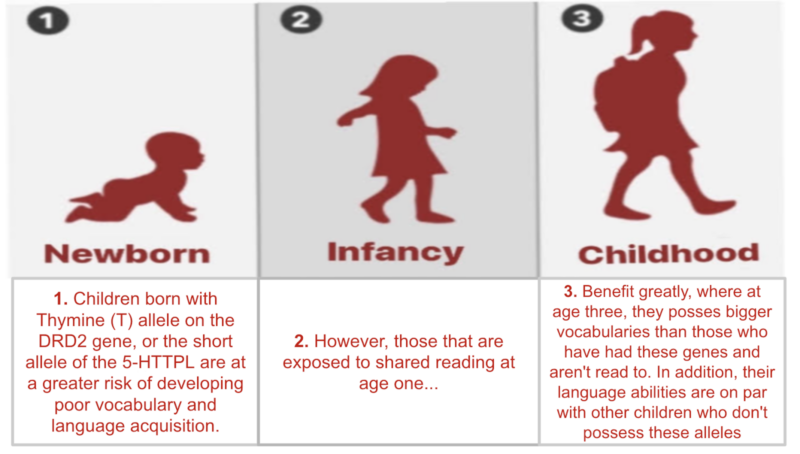
Review written by Sarah McFann (CBE, G5)
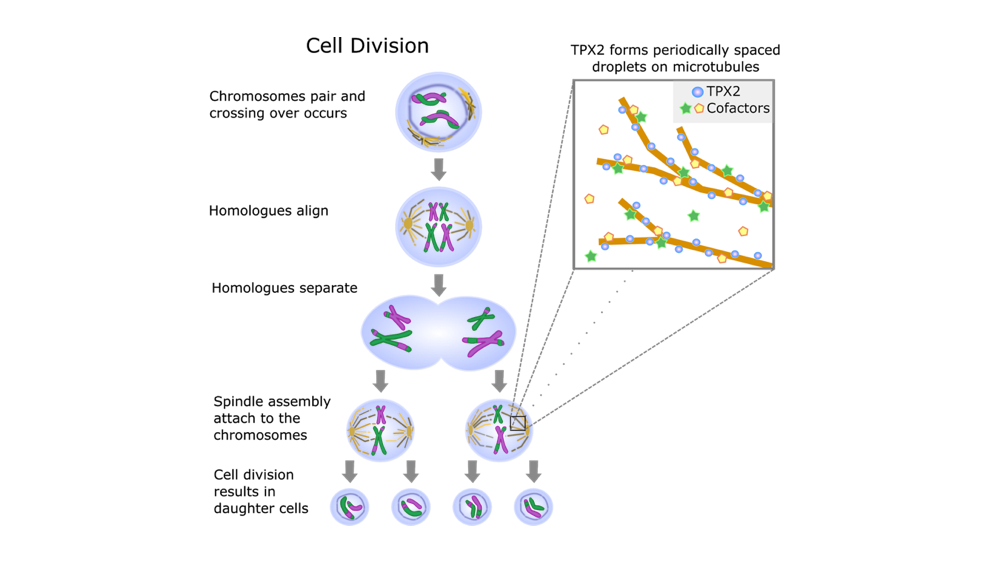
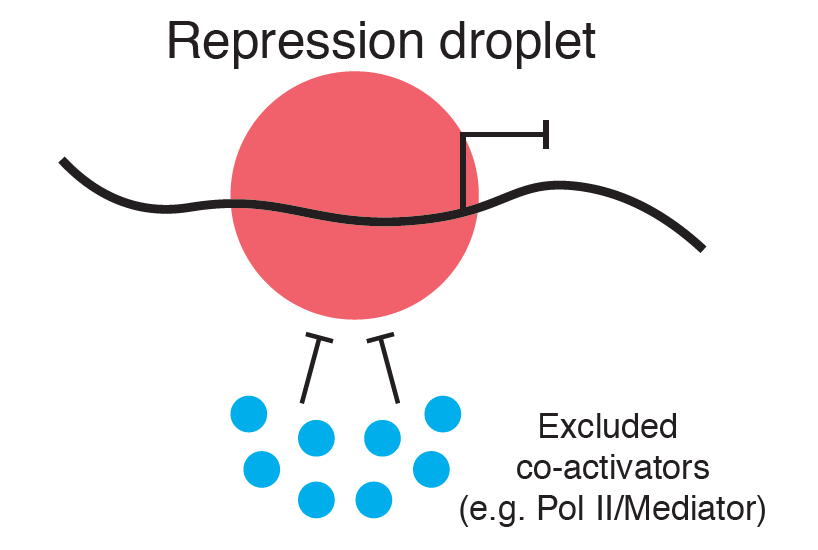
Language is ever-evolving. With each new generation, language structures such as word pronunciation, usage, and meaning mutate and change as they are passed imperfectly from parent to child. Similarly, bodies have the chance to evolve with every generation. Mutations in the germ line—eggs in females and sperm in males—give rise to the genetic variation that allows form and function to evolve. With each new germline mutation, the nucleotides that make up the genetic code are altered due to imperfect DNA replication. These mutations can code for changes to protein structures or protein amounts, altering the way bodies are constructed as they develop and the forms they take on along the way.
Continue reading