Review written by Sarah McFann (CBE, G6)
One thing that sets humans apart from our closest evolutionary relatives, Old World apes and monkeys, is that much of human brain development occurs outside the womb. This means that, relative to our evolutionary neighbors, humans are born altricial—a term describing animals that are born helpless and dependent upon parental care. Because our immature brains are presented with real-world stimuli as they develop, humans have the chance to be molded by external cues like language.
Infants learn language from the people that take care of them, and even at a pre-speech age, infant babbling resembles the languages they hear.1,2 Because humans are a prosocial species—a species in which individuals act to benefit others, even when those others aren’t close kin—adults other than an infant’s parents may respond to an infant’s babbling. For example, at a daycare center or a doctor’s office, an infant’s babbling may draw the attention of a childcare worker or a physician who can aid the child. Cooperative breeding is a term used to describe this kind of behavior, in which adults other than an infant’s mother regularly contribute to the infant’s wellbeing.
In a Developmental Psychobiology paper published earlier this year,3 Princeton PhD candidate Thiago Varella and Princeton Professor Asif Ghazanfar ask the question: Is the ability to learn from adult sounds connected to altriciality and cooperative breeding? Their reasoning was that infant humans are energetically demanding to raise because they are born altricial, so they rely upon adults to provide for their needs. When an infant is in need, they attract attention by making noise. Since humans are a prosocial species, any person might respond to the infant’s call, whether they are related to the infant or not. Thus, it serves the infant to make noises that adults respond to in general. Because adults are more likely to respond to infants whose babbling sounds more speechlike,4 mimicking adult sounds could work as a strategy for drawing a caregiver’s attention.
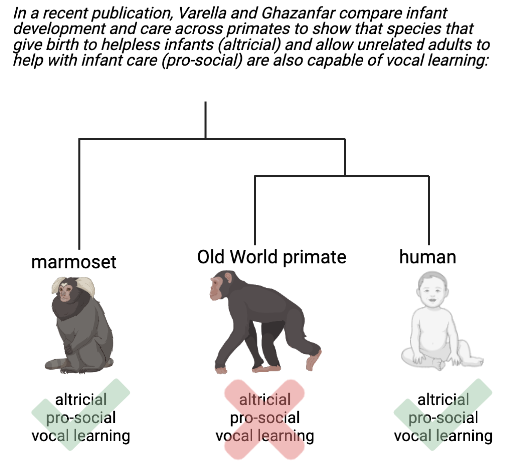
Varella and Ghazanfar explored this question by looking at how these three traits—altriciality, cooperative breeding, and vocal learning—relate to one another in non-human species. Our closest evolutionary neighbors, the Old World monkeys and apes, do not share any of the three traits with us. Old World primates give birth to infants that are significantly more independent than human infants. They also don’t use cooperative breeding strategies. Only in rare situations will a mother allow another to hold her child, and only then in exchange for grooming or time at a feeding site. Moreover, unrelated individuals don’t carry infants or provide food for them in order to be helpful. Finally, Old World primates are born already making mature-sounding vocalizations, so they aren’t considered vocal learners.
In light of this, the species Varella and Ghazanfar settled on for their analysis was marmoset monkeys. Marmosets are New World monkeys that branched off from humans 40 million years ago— 15 million years before humans branched from Old World primates.5 Like humans, marmosets are born altricial, but due to a different cause. Human embryos are energetically demanding to carry to term due to the high energy needs of the developing infant brain. Marmoset births, on the other hand, are energetically costly because marmosets are obligate producers of twins, meaning they almost always give birth to at least two infants per pregnancy (humans, by contrast, only produce twins 3% of the time6). To compensate for the energetic demands of carrying twins, marmosets have evolved a strategy in which organ development, including brain development, is stalled in the womb. This leads to marmosets being born smaller and less developed than other nonhuman primates.
Marmosets are also prosocial and use cooperative breeding strategies to care for their infants. Marmoset mothers regularly pass their infants to other adult marmosets, demonstrating that, unlike Old World primates, marmosets are routinely willing to let other members of their species help with infant care. Adult marmosets also readily share food with infants, even unrelated ones.7
Marmoset vocal learning trends also overlap well with human trends. Adult marmosets make a long, loud, high-pitched “phee” sound to contact other marmosets that are out of sight—a sound that newborns have trouble returning. By two months of age, however (which corresponds to about two years of age in humans), marmosets can reliably replicate the call. Varella and Ghazanfar noted that an infant marmoset’s ability to make the “phee” call depends on how often its caregivers communicate with it. The more often adult marmosets respond to an infant marmoset’s immature call with a correct call, the faster the infant learns how to replicate it.
Combining these observations together, Varella and Ghazanfar concluded that altriciality, cooperative breeding, and vocal learning are evolutionarily linked. Both humans and marmosets exhibit all three traits together, while Old World primates do not—at least not nearly to the same degree. It is likely these traits arose in both humans and marmosets as a product of convergent evolution, which happens when organisms that are not closely related evolve similar traits as a result of adapting to similar environments.
Varella and Ghazanfar acknowledge Princeton’s animal facilities for making their research possible. “Princeton provided state-of-the-art housing facilities and veterinary care for our small colony of marmoset monkeys,” said Varella and Ghazanfar. “Using this colony, we were able to demonstrate, for the first time, vocal learning in a primate. This finding, of course, opened the door to thinking about why (as far as we know) only marmosets and humans exhibit this behavior.”
Varella and Ghazanfar’s study highlights how traits like cooperative breeding and the ability to learn based on social interactions are not uniquely human. Humans and marmosets share these similarities despite significant differences in species-specific factors like brain size. Rather than being a unique feature of the human brain, our prosocial tendencies and ability to adjust the way we communicate based on the way others communicate with us appears to be rooted in our leaving the womb at a relatively undeveloped stage.
Moving forward, Varella and Ghazanfar are interested in building models of vocal learning across species. “We no longer work with marmosets, or any other animals, but have a very large data set of their infant and adult vocal behaviors which we are using to build dynamical systems models of vocal development,” Varella and Ghazanfar explained. “For example, a recent study investigates how the early vocal development of three different mammals (marmoset monkeys, Egyptian fruit bats, and humans) undergo at least one sudden transition in the acoustics of their developing vocalizations. To understand the mechanism, we modeled different developmental landscapes and showed that the natural dynamics of these landscapes are the result of changes in energy expenditure and information transmission over the course of infant development.”
Many questions surrounding the origin of human communication remain unanswered, like why humans are the only species to possess complex language, or what role biological evolution versus cultural evolution played in the emergence of human language. By drawing comparisons between human and marmoset development, social structures, and communication, Varella and Ghazanfar’s study provides a new perspective on how genetics, social interactions, and brain development work in tandem to influence the ways in which we learn to communicate.
References
- Mampe, B., Friederici, A. D., Christophe, A., & Wermke, K. (2009). Newborns’ cry melody is shaped by their native language. Current biology, 19(23), 1994-1997.
- Wermke, K., Teiser, J., Yovsi, E., Kohlenberg, P. J., Wermke, P., Robb, M., … & Lamm, B. (2016). Fundamental frequency variation within neonatal crying: Does ambient language matter?. Speech, Language and Hearing, 19(4), 211-217
- Varella, T. T., & Ghazanfar, A. A. (2021). Cooperative care and the evolution of the prelinguistic vocal learning. Developmental Psychobiology.
- Gros-Louis, J., West, M. J., Goldstein, M. H., & King, A. P. (2006). Mothers provide differential feedback to infants’ prelinguistic sounds. International Journal of Behavioral Development, 30(6), 509-516
- Preuss, T. M. (2016). Primate brain evolution. The International Encyclopedia of Primatology, 1-4.
- Martin, J. A., Hamilton, B. E., Osterman, M. J. K., Driscoll, A. K. (2021). Births: Final Data for 2019. National Vital Statistics Reports, 70(2). Hyattsville, MD: National Center for Health Statistics. DOI: https://dx.doi.org/10.15620/cdc:100472.
- Burkart, J. M., Hrdy, S. B., & Van Schaik, C. P. (2009). Cooperative breeding and human cognitive evolution. Evolutionary Anthropology: Issues, News, and Reviews: Issues, News, and Reviews, 18(5), 175-186.