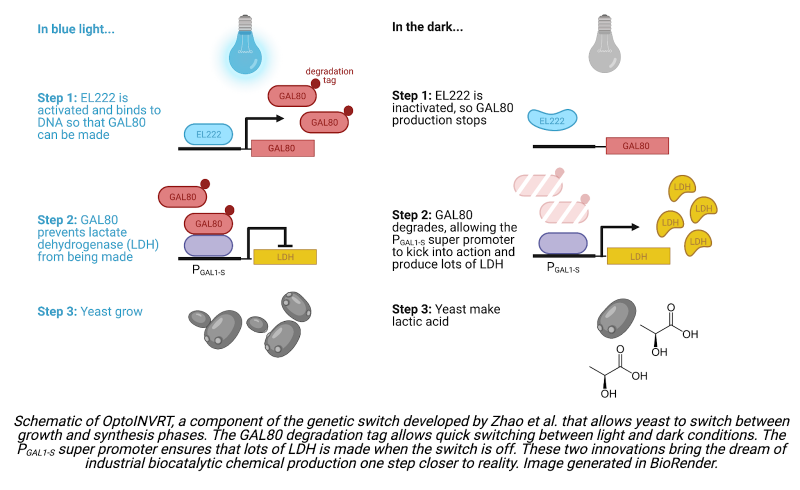
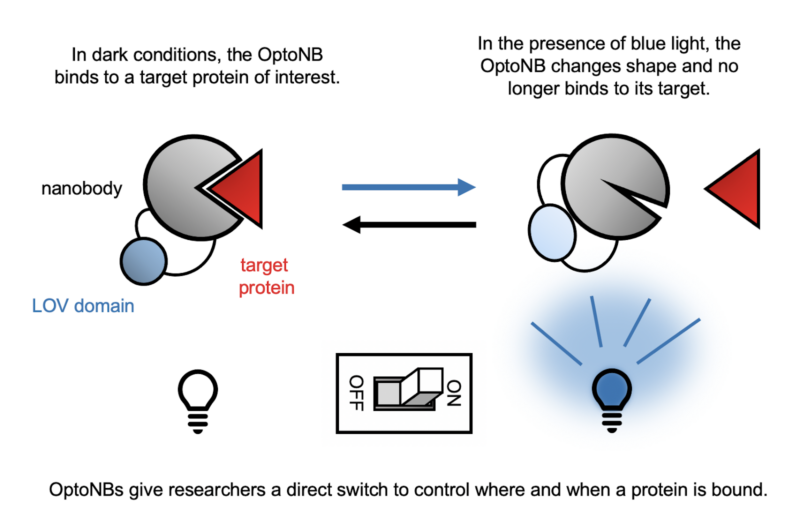
Review written by Sarah McFann (CBE, G5)
Many household goods, from dyes and plastics to contact lenses and aspirin, are made using petroleum byproducts. Over the past 150 years, chemical catalysts have been optimized to efficiently convert crude oil into starting materials for a wide range of products. Unfortunately, petroleum is a non-renewable resource, and emissions from petroleum processing are a big contributor to climate change. A team of bioengineers from the Avalos Lab at Princeton University is investigating an alternative: a petroleum-free way of manufacturing carbon-based goods that uses genetically engineered yeast to convert sugar into high-value products.
Continue reading